|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Preparation.
Sodium hypochlorite (NaClO) and sodium chloride (NaCl) are formed
when chlorine is passed into cold and dilute sodium hydroxide
solution. It is prepared industrially by electrolysis with minimal
separation between the anode and the cathode. The solution must be
kept below 40 °C (by cooling coils) to
prevent the undesired formation of sodium chlorate (see Noted,
hereunder).
Hence, chlorine is simultaneously reduced and oxidized; this
process is known as disproportionation.
Commercial solutions always contain significant amounts of sodium
chloride (common salt) as the main by-product, as seen in the
equation above.
The concentrations obtained, are 12.5 to 25% chlorine
(in the latter case, partially precipitated NaCl and
must be removed).
Noted.
Parasitic reaction to slow as possible: NaClO 3 >>> NaClO 3
+ 2 NaCl, that is to say, the formation of chlorates and chlorides
NaClO3, this is made possible by maintaining the temperature of the
environment as low as possible (do not exceed 40
° C).
Reaction of calcium chloride on sodium carbonate:
(calcium carbonate CaCO3 precipitates)
Notes:
The pure sodium hypochlorite in the form of solid crystals (hydrated
in variable values) is of little interest due to the low stability of
this substance. it is more stable in cold dilute solutions.
Production.
FRENCH SITUATION: production, about 245 million liters / year in
public (which is twice the volume of household cleaners).
Every business day, using about 1 million of concentrated bleach
cartons.
France is the second rank of European consumption by volume, behind
Spain and ahead of Italy (about 220 million liters / year).
In addition, France is the 5th largest global consumption volume
behind:
Title of chlorine (liquid bleach):
The French chlorometric degree ° Cl (definition still used
primarily in French-speaking countries):
This is the oxidizing power of a liter of bleach at 20 °
C, expressed in liters of dry chlorine gas at 1
bar (1013 hPa) and at 0 ° C.
It corresponds to chlorine gas Cl2 used to make bleach.
Or at T = 20 °C, and P = 1 bar, 1 liter Cl2,3.17 g in weigh
(44.7 mmol), thus
(Reminder: 1 Cl = 3.32 g NaClO / liter [3.32 gL]).
THIS UNIT HAS BEEN ABANDONED IN 2001 - THE FRENCH WATER BLEACH SHOULD
NOW BE IN% OF ACTIVE CHLORINE.
Note: in some countries, Cl ° is defined as the number of liters
of chlorine gas Cl2 (at 0 ° C and 760 mm Hg), contained in 1
liter of solution; So in this case ° Cl = 3.214 g active Cl2 /
l.
Percent active chlorine.
Percent active chlorine is a unit of concentration used for
hypochlorite-based bleaches. One gram of a 100% active chlorine
bleach has the same bleaching power as one gram of chlorine. The term
"active chlorine" is used because most commercial bleaches contain
substantial amounts of chlorine in the form of chloride ions, which
have no bleaching properties.
This definition as a bleach (Anglo-Saxon) was adopted at the European
level in 1994. It reports the total amount of chlorine Cl2 used in
the manufacture of bleach.
However, the% of active chlorine, for the same quality of bleach,
depends on the density of the bleach which itself varies with the
preparation method of the bleach.
Indeed, bleach dilution prepared by bleach at 100 ° Cl is less
dense (a portion of ions Na + and Cl-was removed) a bleach prepared
directly.
The chlorine term is - despite its widespread use in Europe -
improper ...
Rather chemists employ the term "available chlorine".
Note also that an excess of ions HO-(5 to 12 g / l expressed as NaOH)
is held in the NaClO solution in order to neutralize the influence of
CO2 from the air. As a result, the pH of a concentrated bleach can be
strongly basic (pH = 11.5).
It is therefore necessary to know the relationship between density
and concentration to convert ° Cl%, which moreover may vary,
depending on the raw materials used because there are two different
materials:
(sources -French) : Chambre Syndicale de l'Eau de Javel)
The tables below, collect data for conversion between units, and allow you to perform calculations for dilute solutions.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Table
(pdf, 53 KB, in French) to download (CSNEJ -
05/2010).
Commercial products.
Until 1976, the bleach was stabilized and colored orange with sodium
dichromate (about 20 g per 100 liters).
Currently, the commercial bleach is a clear, yellow-green, with a
characteristic smell called "chlorinated" and between 1.0 and 1.2
average density.
Solutions are marketed under two main forms:
The concentrated extract at 35 ° Cl is never used pure but diluted quart (250 ml + 750 ml concentrated bleach distilée of water). This results in a bleach grading 8.75 ° Cl about.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Properties (equimolar solution of [NaClO + NaCl], at
50 ° Cl):
Other features: :
Oxidizing properties: it is more oxidizing than its pH is low, but
even at pH 14 its oxidizing power remains high (E ° = 0.88
V).
It can oxidize many toxic compounds in "harmless" compounds, such as
for example:
Its bleaching action in part, due to its ability to oxidize many organic compounds.
Disinfectant properties:
These are due to the bactericidal action of hypochlorous acid which
diffuses through the cell wall of bacteria destroying membrane
proteins.
Furthermore, HClO acts on the metabolism of bacterial synthesis.
HClO, unloaded, is nearly 100 times more bactericidal than the
hypochlorite ion.
In the case of virus attack by HClO would amidated bonds of
proteins.
At the request of the National Union Chamber of Bleach
(Chambre Syndicale Nationale de l'Eau de Javel,
CSNEJ), the Pasteur Institute of Lille in 2008 an essay based
on the European standard EN 14476 to determine the virucidal activity
of the water bleach to 2.6% active chlorine on the Influenza A/H5N1
virus.
The results show that the bleach has virucidal activity on avian
influenza A/H5N1.
Under the following conditions:
The effective concentration is 0.10% chlorine.
This corresponds to the bleach bottle of commerce 25 times
diluted about.
Chemical properties.
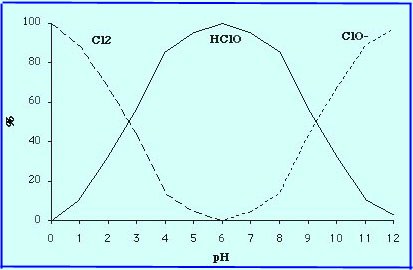
When a sodium hypochlorite
[Na+][ClO-] solution is
injected in water in pH values (5.5 to 9) normally encountered water,
all or part of the ion [ClO-] is transformed into
hypochlorous acid HClO :
Note therefore that 1 millimole hypochlorite "consumes" 1 meq
CO2, and generates 1 meq alkalinity
(TAC), which would increase the pH slightly
(some water).
Chlorinated forms depending on pH:

Thus, sodium hypochlorite has the same actions as the chlorine
gas.
Stability :
Uses.
Bleaching.
Household bleach is, in general, a solution containing 3-8%
sodium hypochlorite and 0.01-0.05% sodium hydroxide; the sodium
hydroxide is used to slow the decomposition of sodium hypochlorite
into sodium chloride and sodium chlorate.
In household form, sodium hypochlorite is used for removal of stains
from laundry. It is particularly effective on cotton fiber, which
stains easily but bleaches well. Usually 50 to 250 mL of bleach per
load is recommended for a standard-size washer. The properties of
household bleach that make it effective for removing stains also
result in cumulative damage to organic fibers, such as cotton, and
the useful lifespan of these materials will be shortened with regular
bleaching. The sodium hydroxide (NaOH) that is also found in
household bleach (as noted later) causes fiber degradation as well.
It is not volatile, and residual amounts of NaOH not rinsed out will
continue slowly degrading organic fibers in the presence of humidity.
For these reasons, if stains are localized, spot treatments should be
considered whenever possible. With safety precautions, post-treatment
with vinegar (or another weak acid) will neutralize the NaOH, and
volatilize the chlorine from residual hypochlorite. Old T-shirts and
cotton sheets that rip easily demonstrate the costs of laundering
with household bleach. Hot water increases the effectiveness of the
bleach, owing to the increased reactivity of the molecules.
Stain removal.
Sodium hypochlorite has destaining properties.[11]
Amongst other applications, it can be used to remove mold stains,
dental stains caused by fluorosis,[12] and stains on
crockery, especially those caused by the tannins in tea.
Disinfection.
A weak solution of 2% household bleach in warm water is used
to sanitize smooth surfaces prior to brewing of beer or wine.
Surfaces must be rinsed to avoid imparting flavors to the brew; the
chlorinated byproducts of sanitizing surfaces are also harmful. The
mode of disinfectant action of sodium hypochlorite is similar to that
of hypochlorous acid.
A 1-in-5 dilution of household bleach with water (1
part bleach to 4 parts water) is effective against many
bacteria and some viruses, and is often the disinfectant of choice in
cleaning surfaces in hospitals (primarily in the
United States). The solution is corrosive and needs to be
thoroughly removed afterwards, so the bleach disinfection is
sometimes followed by an ethanol disinfection. Liquids containing
sodium hypochlorite as the main active component are also used for
household cleaning and disinfection, for example toilet cleaners.
Some cleaners are formulated to be thick so as not to drain quickly
from vertical surfaces, such as the inside of a toilet bowl.
Deodorising.
Sodium hypochlorite has deodorising properties.
Endodontics.
Sodium hypochlorite is now used in endodontics during root
canal treatments. It is the medicament of choice due to its efficacy
against pathogenic organisms and pulp digestion. In previous times,
Henry Drysdale Dakin's solution (0.5%) had been used. Its
concentration for use in endodontics today varies from 0.5% to 5.25%.
At low concentrations it will dissolve mainly necrotic tissue;
whereas at higher concentrations tissue dissolution is better but it
also dissolves vital tissue, a generally undesirable effect. It has
been shown that clinical effectiveness does not increase conclusively
for concentrations higher than 1%.
Nerve agent neutralization.
At the various nerve agent (chemical warfare nerve gas) destruction
facilities throughout the United States, 50% sodium hypochlorite is
used as a means of removing all traces of nerve agent or blister
agent from Personal Protection Equipment after an entry is made by
personnel into toxic areas. 50% sodium hypochlorite is also used to
neutralize any accidental releases of nerve agent in the toxic areas.
Lesser concentrations of sodium hypochlorite are used in similar
fashion in the Pollution Abatement System to ensure that no nerve
agent is released in furnace flue gas.
Reduction of skin damage.
Dilute bleach baths have been used for decades to treat moderate to
severe eczema in humans,[15][16] but it has not been
clear why they work. According to work published by researchers at
the Stanford University School of Medicine in November 2013, a very
dilute (0.005%) solution of sodium hypochlorite in water was
successful in treating skin damage with an inflammatory component
caused by radiation therapy, excess sun exposure or ageing in
laboratory mice. Mice with radiation dermatitis given daily 30-minute
baths in bleach solution experienced less severe skin damage and
better healing and hair regrowth than animals bathed in water. A
molecule called nuclear factor kappa-light-chain-enhancer of
activated B cells (NF-kB) is known to play a critical role in
inflammation, ageing and response to radiation. The researchers found
that if NF-kB activity was blocked in elderly mice by bathing them in
bleach solution, the animals' skin began to look younger, going from
old and fragile to thicker, with increased cell proliferation. The
effect diminished after the baths were stopped, indicating that
regular exposure was necessary to maintain skin thickness.
Water treatment.
In drinking water systems, swimming pools, etc., sodium
hypochlorite is widely used for chlorination. Hypochlorites are an
alternative to chlorine gas, which is difficult to handle in many
contexts. Also, accidents involving chlorine gas are more serious
than accidents involving hypochlorites. Chlorination usually produces
small quantities of harmful byproducts. Hypochlorites are very
similar to chlorine gas in this regard.
Sodium hypochlorite solutions have been used to treat dilute cyanide
wastewater, such as electroplating wastes. In batch treatment
operations, sodium hypochlorite has been used to treat more
concentrated cyanide wastes, such as silver cyanide plating
solutions. Toxic cyanide is oxidized to cyanate (CNO-)
that is not toxic, idealized as follows:
Sodium hypochlorite is commonly used as a biocide in industrial
applications to control slime and bacteria formation in water systems
used at power plants, pulp and paper mills, etc. in solutions
typically of 10-15% by weight.
The following comparative table shows the characteristics of the
bleach (or chlorine) and ozone :
|
|
|
|
|
virus: 1 amoebae: 0,05 ---- duration : 45 min |
virus : 5 amoebae : 0,5 ---- duration : 4 min |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
At lower concentrations lethal doses, bleach inhibits the growth
of bacteria, thus protecting the drinking water during transportation
by pipeline.
Neutralization.
Sodium thiosulfate is an effective chlorine neutralizer. Rinsing
with a 5 mg/L solution, followed by washing with soap and water,
quickly removes chlorine odor from the hands.
Storage :
Note that sodium hypochlorite is preserved even better than the
temperature is lower.
As a result of degassing of chlorine to avoid
désamorçages metering pumps distribution vents are
provided on the suction and discharge lines.
It can be delivered by carboys, containers or tankers.
Ecological Information: Do not allow to enter waters, waste
water or soil!
Safety.
Sodium hypochlorite is a strong oxidizer. Oxidation reactions are
corrosive, solutions burn skin and cause eye damage, especially when
used in concentrated forms. However, as recognized by the NFPA, only
solutions containing more than 40% sodium hypochlorite by weight are
considered hazardous oxidizers. Solutions less than 40% are
classified as a moderate oxidizing hazard (NFPA 430, 2000).
Mixing bleach with some household cleaners can be hazardous. For
example, mixing an acid cleaner with sodium hypochlorite bleach
generates chlorine gas. Mixing with ammonia solutions (including
urine) produces chloramines. Mixtures of other cleaning agents and or
organic matter can result in a gaseous reaction that can cause acute
lung injury.
Both chlorine gas and chloramine gas are toxic. Bleach can react violently with hydrogen peroxide and produce oxygen gas:
It is estimated that there are about 3300 accidents needing
hospital treatment caused by sodium hypochlorite solutions each year
in British homes (RoSPA, 2002).
Household bleach and pool chlorinator solutions are typically
stabilized by a significant concentration of lye (caustic soda, NaOH)
as part of the manufacturing reaction. Skin contact will produce
caustic irritation or burns due to defatting and saponification of
skin oils and destruction of tissue. The slippery feel of bleach on
skin is due to this process. Trichloramine, the gas that is in
swimming pools can cause atopic asthma.
A recent European study indicated that sodium hypochlorite and
organic chemicals (e.g., surfactants,
fragrances) contained in several household cleaning products
can react to generate chlorinated volatile organic compounds (VOCs).
These chlorinated compounds are emitted during cleaning applications,
some of which are toxic and probable human carcinogens. The study
showed that indoor air concentrations significantly increase
(8-52 times for chloroform and 1-1170 times for
carbon tetrachloride, respectively, above baseline quantities in the
household) during the use of bleach containing products. The
increase in chlorinated volatile organic compound concentrations was
the lowest for plain bleach and the highest for the products in the
form of “thick liquid and gel”. The significant increases
observed in indoor air concentrations of several chlorinated VOCs
(especially carbon tetrachloride and chloroform) indicate that the
bleach use may be a source that could be important in terms of
inhalation exposure to these compounds. The authors suggested that
using these cleaning products may significantly increase the cancer
risk.
Chlorination of drinking water can oxidize organic contaminants,
producing chloroform and other trihalomethanes, which are
carcinogenic, and many hundreds of possible disinfection by-products,
the vast majority of which are not monitored.
One major concern arising from sodium hypochlorite use is that it
tends to form chlorinated organic compounds; this can occur during
household storage and use as well during industrial use. For example,
when household bleach and wastewater were mixed, 1-2% of the
available chlorine was observed to form organic compounds. As of
1994, not all the byproducts had been identified, but identified
compounds include chloroform and carbon tetrachloride. The estimated
exposure to these chemicals from use is estimated to be within
occupational exposure limits.
COMPOUNDS CONTAINING ION HYPOCHLORITES:
There are compounds containing hypochlorite ions in solid
form.
It is primarily:
 (use your browser)
(use your browser)