Aluminium sulfate,
alternatively spelled either
aluminum or
sulphate.
Other names: Cake alum, Filter alum, Papermaker's alum,
Alunogenite.
Empirical formula (anhydrous) :
Al2(SO4)3
Molar masses :
Al2(SO4)3 = 342.15
g·mol-1(anhydrous)
(O = 56,11 %, S = 28,12 %, Al = 15,77
%)
Al2(SO4)3, 18 H2O =
666.42 g·mol-1
(octadecahydrate)
Al2(SO4)3, 14 H2O = 594
g·mol-1
Aluminium sulfate is sometimes referred to as a type of alum. Alums
are double sulfate salts, with the formula
AM(SO4)2·12H2O, where A is a
monovalent cation such as potassium or ammonium and M is a trivalent
metal ion such as aluminium. The anhydrous form occurs naturally as a
rare mineral millosevichite, found e.g. in volcanic environments and
on burning coal-mining waste dumps. Aluminium sulfate is rarely, if
ever, encountered as the anhydrous salt. It forms a number of
different hydrates, of which the hexadecahydrate Al2(SO4)3•16H2O
and octadecahydrate
Al2(SO4)3•18H2O are
the most common. The heptadecahydrate, whose formula can be written
as [Al(H2O)6]
2(SO4)3•5H2O, occurs naturally
as the mineral alunogen.
Preparation.
Aluminium sulfate may be made by adding aluminium hydroxide,
Al(OH)3, to sulfuric acid,
H2SO4:
It is available in various solid forms (depending on the manufacturer):
Main features :
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|

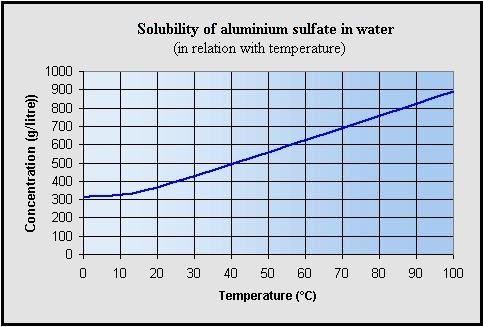
The insoluble portion is maximum 0.3% (w / w).
The solutions are to be stored (preferably)
above 0 ° C.
Chemical action.
The chemical action of conventional coagulants gives:
A formulation for water
treatment.
Aluminium sulfate react with dissolved calcium
bicarbonate.
12
Al2[SO4]3,14H2O...+
...3
Ca[HCO3]2
.............>>>>>>....
2 Al[OH]3 ...+
...6CO2.....+....3CaSO4
........(342+252)=594.................(3x162)=486
or 30°F as TAC v
...........(2x78)=156............(6x44)=264.........(3x96
as SO4)=288
Par mg/L aluminium sulfate added there (30/594) = 0.05 °F
lower Alkalinity (TAC), and is formed
(264/594) = 0.444 mg of free CO2, and is also formed (156/594) =
0.263 mg of aluminum hydroxide (as insoluble
flocs), and (288/594) = 0.485 mg of dissolved sulphate.
Standardization.
Standard products used for the production of drinking
water: Official
French Bulletin - Aluminium sulfate : NF EN 878.
Uses.
Safety.
Alzheimer's disease:
There have been studies that have found an association between
exposure to and long-term use of antiperspirants and Alzheimer's
disease, however the studies also have shown that the association is
negligible (less than 1%). There is no adequate evidence that
exposure to aluminium in antiperspirants leads to progressive
dementia and Alzheimer's disease.
Breast cancer:
The International Journal of Fertility and Women's Medicine found no
evidence that certain chemicals used in underarm cosmetics increase
the risk of breast cancer. Ted S. Gansler, MD, MBA, the director of
medical content for the American Cancer Society, stated "There is no
convincing evidence that antiperspirant or deodorant use increases
cancer risk".
The European Journal of Cancer Prevention stated "underarm shaving
with antiperspirant/deodorant use may play a role in breast cancer.
"The journal Breast Cancer Research proposed a link between breast
cancer and the application of cosmetic chemicals in the underarm,
including aluminium, with oestrogenic and/or genotoxic properties.
Personal care products are potential contributors to the body burden
in aluminium and newer evidence has shown that more aluminium is
deposited in the outer regions than the inner regions of the breast.
But whether differences in the distribution of aluminium are related
to higher incidence of tumours in the outer upper region of the
breast remains unknown.
Effects on the environment.
Aluminum sulfate may decrease the pH of stream, thus representing
a potential threat to aquatic fauna and flora
(stream, river, groundwater).
Sources : personal and Wikipedia,
the free encyclopedia.
 (use your browser)
(use your browser)